Antibiotic-resistant bacteria have become a major global health threat that continues to rise. People of all ages are affected, but pediatric patients and aged individuals are at increased risk of antibiotic-resistant bacterial infections. Vaccines and antibody immunotherapies may be suitable alternatives to antibiotics because bacteria are not able to develop resistance against these immunotherapies. Yet, vaccines and therapeutic antibodies are unavailable for several bacterial infections, including most antibiotic-resistant bacteria. In a new study published by Advanced Functional Materials, Ankur Singh, a recently hired associate professor with joint appointments in the Wallace H. Coulter Department of Biomedical Engineering at Georgia Tech and Emory University, and the Woodruff School of Mechanical Engineering, has developed an immune organoid technology, using synthetic polymers, that explains the immune response against components of a deadly antibiotic-resistant bacteria, Klebsiella pneumoniae, in young and aged mice.
According to Singh, “for us to develop effective vaccination or ex vivo antibodies that can be infused into patients, we need to understand how different parts of bacteria or viruses react with the immune cells that make antibodies to protect us. This has remained a black-box for the scientific field because unlike the human body, there are no suitable technologies to systematically study these [bacterial] components, especially in the case of aging.”
The human body makes antibodies against bacterial and viral antigens through a controlled immunological process, germinal center, in the lymph nodes. This process, much like a Darwinian approach, relies on the tissue microenvironment where germinal center evolves, epigenetic underpinnings, and a decision-making process that ultimately decides whether a B cell (involved in triggering an antibody immune response) will secrete robust antibodies by becoming a plasma cell or die. However, the complex polysaccharide nature of bacterial antigens shields them from germinal center formation and therefore mouse models are often inefficient in predicting the response of novel antigens. This situation is particularly concerning in aging where the human body is unable to mount an efficient immune response to bacterial antigens due to immune senescence and defects in T cells.
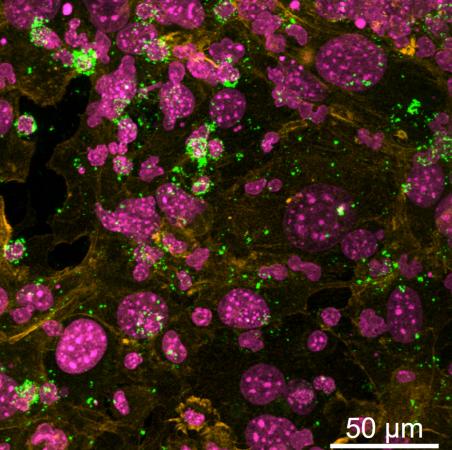
To that end, Singh’s research took a bioinspired approach to rationally develop a synthetic hydrogel organoid of the immune system. In a systematic approach, in collaboration with Jason Cyster, Howard Hughes Medical Institute investigator and a professor at the University of California San Francisco, the team first identified adhesive proteins (genes in this case) in lymph nodes using a single-cell RNA sequencing technology. Guided by these molecular factors, three synthetic polymer-based hydrogel organoids were further developed and tested, including a polyethylene glycol maleimide gel, originally developed at Georgia Tech, to drive the complex process of B cell immunity. The paper is led by a previous postdoctoral fellow in Singh’s laboratory, Pamela Graney.
Klebsiella pneumoniae is one of the most frequent causes of nosocomial infections that causes severe diseases including pneumonia, bacteremia, and urinary tract infection. It has a strong potential to develop antibiotic and drug resistance and is a leading cause of hospital-acquired infections in the United States. However, many grand challenges remain in developing vaccines or immunotherapies against Klebsiella, or for that matter other antibiotic-resistant pathogens as well. Two such challenges are to identify what mode of presentation of antigen can regulate B cell immune response more robustly for antibody generation and whether one can predict the response in aged individuals.
The synthetic immune organoid developed by Singh is the first to form germinal center-like B cells from aged individuals in a dish. The little trick in this study was to present a lymph node-like microenvironment and T cell signal to B cells from aged mice when cultured in organoids, a signal that is otherwise diminished in aged individuals. The current study sets out to demonstrate the effect of K. Pneumoniae antigens in regulating the germinal center-like B cell immunity and its epigenetics, including the histone modification which is critical for antibody immunity in B cells from aged mice that were older than two years in age (>69 years human equivalent). Working in collaboration with Ilana Brito, assistant professor at Cornell University, the study discovered that dangerous antigens that are embedded within the bacterial membrane are less potent than purified protein antigens, opening new possibilities to systematically identify new immunogens and develop antibodies, B cell therapies, and vaccines.
Previous work from Singh’s laboratory, published in Nature Communications and Nature Protocol, has developed a gelatin-nanoparticle-derived immune organoid that demonstrated striking similarities to immunized mouse models. Singh has discussed the potential of developing immune technologies in Nature Reviews Materials and believes the work could be of tremendous use in predicting response against Coronaviruses.
“Although we have recently shown that one can make antigen-specific antibodies using our organoids, much more work is needed to generate plasma and memory cells, and ultimately translate to the human system. The work brings us a step closer and we are excited about the upcoming new opportunities to interface with clinicians and immunologists at Emory University,” says Singh.
Cornell Ph.D. student Kristine Lai and Sarah Post contributed to the study. The study was supported by the National Institute of Allergy and Infectious Diseases of the US National Institutes of Health as well in part by the US National Science Foundation CAREER award, both of which were originally awarded to Ankur Singh at Cornell University and he is continuing the research at the Georgia Institute of Technology.
Media Contact:
Walter Rich
Communications Manager
Wallace H. Coulter Department of Biomedical Engineering
Georgia Institute of Technology
Media Contact
Walter Rich
Keywords
Latest BME News
Jo honored for his impact on science and mentorship
The department rises to the top in biomedical engineering programs for undergraduate education.
Commercialization program in Coulter BME announces project teams who will receive support to get their research to market.
Courses in the Wallace H. Coulter Department of Biomedical Engineering are being reformatted to incorporate AI and machine learning so students are prepared for a data-driven biotech sector.
Influenced by her mother's journey in engineering, Sriya Surapaneni hopes to inspire other young women in the field.
Coulter BME Professor Earns Tenure, Eyes Future of Innovation in Health and Medicine
The grant will fund the development of cutting-edge technology that could detect colorectal cancer through a simple breath test
The surgical support device landed Coulter BME its 4th consecutive win for the College of Engineering competition.